Virus-host interactions and viral pathogenesis
Understanding virus-host interactions and the mechanisms underlying viral pathogenesis is critical to develop effective prevention and control strategies for viral diseases. We are currently working on projects aimed at understanding viral innate immune evasion, infection immunity, viral persistence and pathogenesis. The main focus of our research are emerging viruses including picornaviruses (Senecavirus A and enteroviruses) and coronaviruses (Severe acute respiratory syndrome coronavirus 2, SARS-CoV-2) of animals and humans.
Viral vector and vaccine development
Effective vaccines and vaccine delivery platforms are critical for infection disease prevention and control. We are currently working on the development of novel viral vectors with improved immunogenicity and safety for vaccine delivery in animals. Large DNA viruses are being engineered into safe and highly immunogenic vaccine delivery platforms for use in animals and potentially humans. We are also working on novel DNA and RNA-based vaccine platforms for vaccine delivery in humans and animals.

Virus host-range and host-susceptibility to infection
SARS-CoV-2 is a zoonotic agent that emerged in 2019 and spread throughout the world causing COVID-19 and millions of human deaths. One intriguing feature of SARS-CoV-2 is its broad host range with multiple species being naturally susceptible to infection. Through collaborations with the National Animal Disease Center (NADC) we have demonstrated that white-tailed deer are highly susceptible to SARS-CoV-2, while livestock species such as swine and cattle are not susceptible. We have been working to assess the molecular determinants of SARS-CoV-2 host range and global determinants of host-susceptibility to infection.
From viral genotype-to-phenotype
Understating the effect of genetic signatures that accumulate in viral genomes as they circulate and transmit within hosts or as they spillover and infect new hosts is critical for the design and implementation of effective disease prevention and control measures. We have established genomic surveillance for Senecavirus A and SARS-CoV-2 and have established pipelines to assess the effect of genetic changes in viral pathogenesis, infection dynamics and tissue tropism in vivo. The use of molecular clones coupled with site-directed mutagenesis allows us to investigate the functional effect of these genetic changes using cell culture and/or animal models of infection.
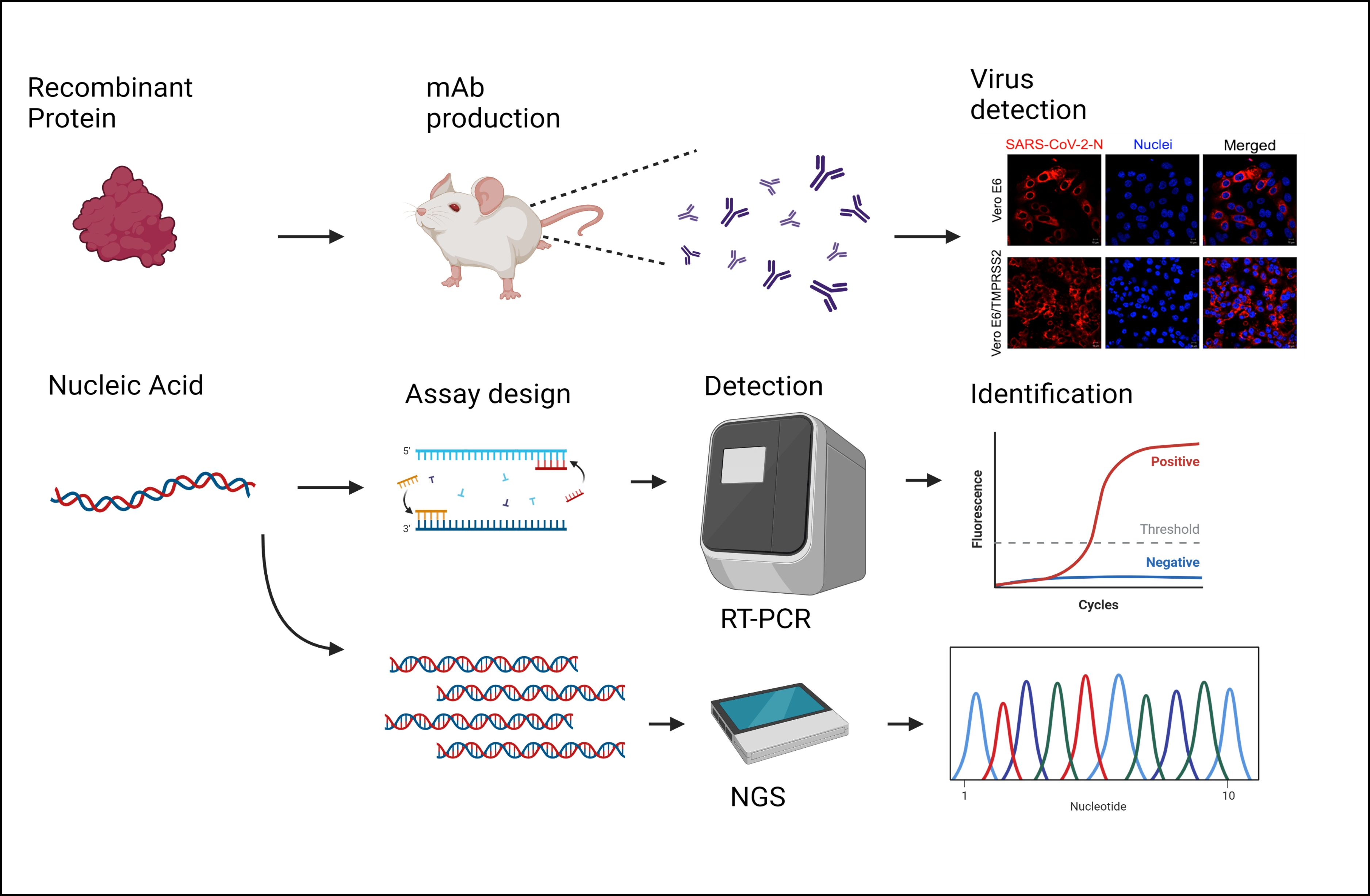
Next generation diagnostics for pathogen detection and identification
Emerging viral diseases pose a constant threat to the humans and animal health. We work to develop novel diagnostic reagents and assays for emerging viral diseases to rapidly isolate, detect and identify emerging viral pathogens. We combine and incorporate classic (i.e. virus isolation, antibody-detection, serological assays, etc) and state-of-the-art techonolgies (i.e next generation sequencing) for viral detection, identification and characterization.
Funding for our research




Learn more
If you have questions or are interested in learning more about our research please send us an e-mail.
